Cataloging the errors and deceptions in the marketing of TMS for adolescent depression
The inaccurate and misleading information about this research is troubling. It's time to correct the record.
I recently did a deep dive into the FDA clearance of TMS for depression in “adolescents” age 15-21. I argued that the data for the NeuroStar device was extremely weak: a negative 103-subject RCT followed by a positive proprietary observational study with major limitations.1
But, what happened next concerns me more than the FDA clearance: a deluge of outright errors, misleading statistics, and frank deception by medical publications and TMS clinics. I may be a jaded psychiatrist, but I am genuinely shocked by how little honest and accurate coverage of the research there is. I worry that families are being misinformed about the data for TMS in adolescent depression.2
Here is my effort to correct the record.
1. TMS websites constantly mistake FDA “clearance” for “approval”
Let’s start with the basics. TMS for adolescent depression was not “FDA approved.” The device was “cleared” for this age range—a far lower bar and a different process than FDA approval. FDA reviews for the approval of medications are typically several hundred pages with detailed analyses of numerous pre-clinical studies and large randomized clinical trials. The NeuroStar FDA clearance packet is 32 pages. The MagVenture one is 18 pages.
But TMS for adolescent depression is constantly referred to as being “FDA approved,” including by the website of the American Academy of Child and Adolescent Psychiatry and countless TMS clinic websites marketing the treatment.
2. Descriptions of the number of adolescents in the FDA study are wrong—by 100 fold
Multiple psychiatrists and TMS clinics have described the FDA dataset as including 10,000 “kids” or “adolescents,” as in:
“The FDA label, TMS treatment as an adjuvant to first-line treatment for depression, which, remember, includes psychotherapy, is down to age 15, and that approval was based on post-marketing data in the real world at a massive scale. So, we’re talking about 10,000 kids who were submitted to the FDA for that post-marketing data set.” [Link, my emphasis]
Or:
“For the FDA study, the outcomes of close to 10,000 adolescents who were treated with Neurostar TMS were examined (source: Neurostar).” [Link]
Or:
The FDA’s approval came after examining outcomes from nearly 10,000 adolescents treated with TMS therapy. [Link]
But this is egregiously inaccurate—the actual dataset submitted to the FDA included 1,169 patients, roughly 90% of whom were 18 or older, as in the figure below. This is wrong by an order of magnitude, and probably several orders of magnitude in reference to “kids” in the dataset.3 Remember, the published data included two 12 year olds, two 13 year olds, seven 14 year olds, and seventeen 15 year olds. How on earth did this become 10,000 “kids?”
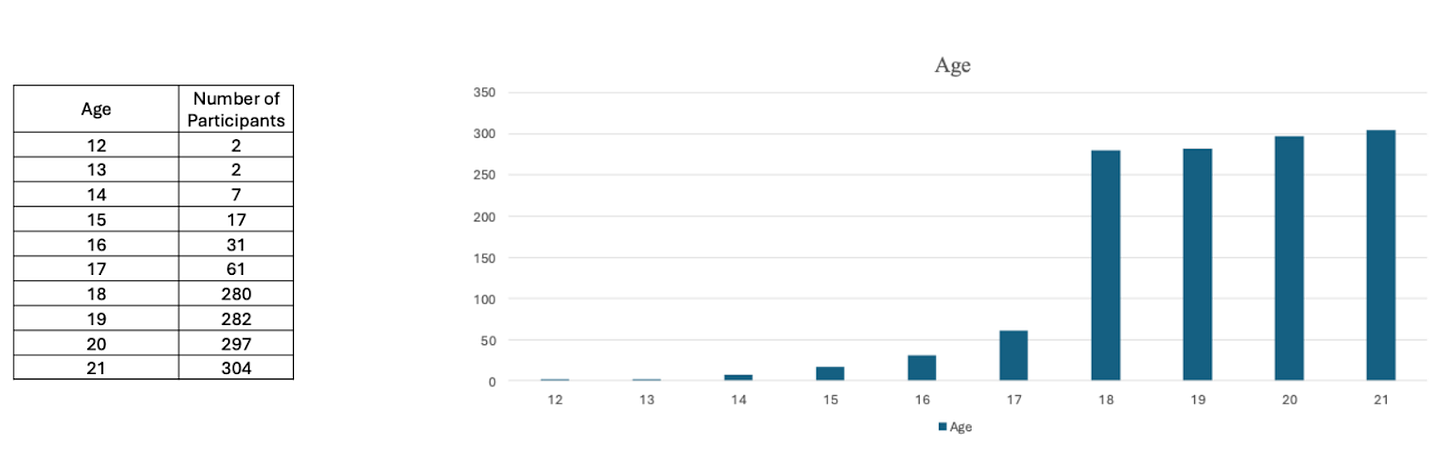
Other pieces in Psychiatric Times and Contemporary Pediatrics suggest that the study included large numbers of younger adolescents by describing the included age range as 12 to 19 years. While factually accurate, this reporting disguises the fact that only four patients were ages 12 and 13. Only 28 patients were under 16! There were 140 times more 19-year olds than 12-year olds in the study.
This basic information on the age distribution is not presented. Readers would come away with a distorted understanding of the data for younger adolescents. Even the FDA recognized the absurdity of clearing a device based on two 12-year olds, hence the clearance down to age 15.4
I can’t fault the NeuroStar manufacturer from including two 12 year-olds in the study so they could describe the research as being on 12 to 21 year-olds and disguise the actual age distribution of patients. They are simply trying to make money. As the CEO stated in their press release about the adolescent clearance: “As a company, we will be focused on driving even more awareness and education about NeuroStar given that this new clearance grows our total addressable market in MDD by 35%.”
But where are the journalists, reviewers, or researchers to point out how few younger adolescents were actually studied?
3. Descriptions of the outcomes are misleading and deceptive
Countless TMS clinic websites and several healthcare publications report the same outcomes for the FDA study: a 78% response rate and 48% remission rate. These numbers are truly impressive. What psychiatrist or parent wouldn’t be enthusiastic?
I suspect that they are informed by the NeuroStar website which advertises these data, paired, coincidentally, with a statement that 10,000 adolescents have been treated.
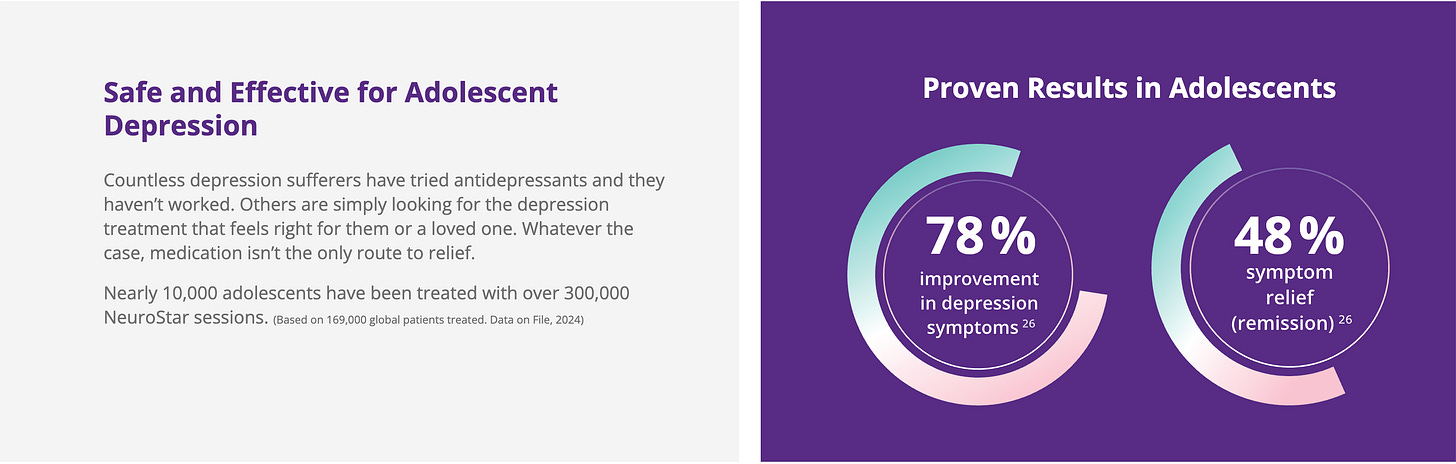
The problem is that these outcomes are utterly misleading. These weren’t the primary outcomes for the 1,169 patients in the study, which were a response rate of 56% and a remission rate of 28% on the Patient Health Questionnaire-9.5
The 78% and 48% outcomes come from a secondary analysis of only 77 patients (6.6% of the sample) who also had a Clinical Global Impressions-Severity score.6 Only 42 of the 77 patients were “adolescents,” up to age 19: outcomes put in the Supplementary Table 4.
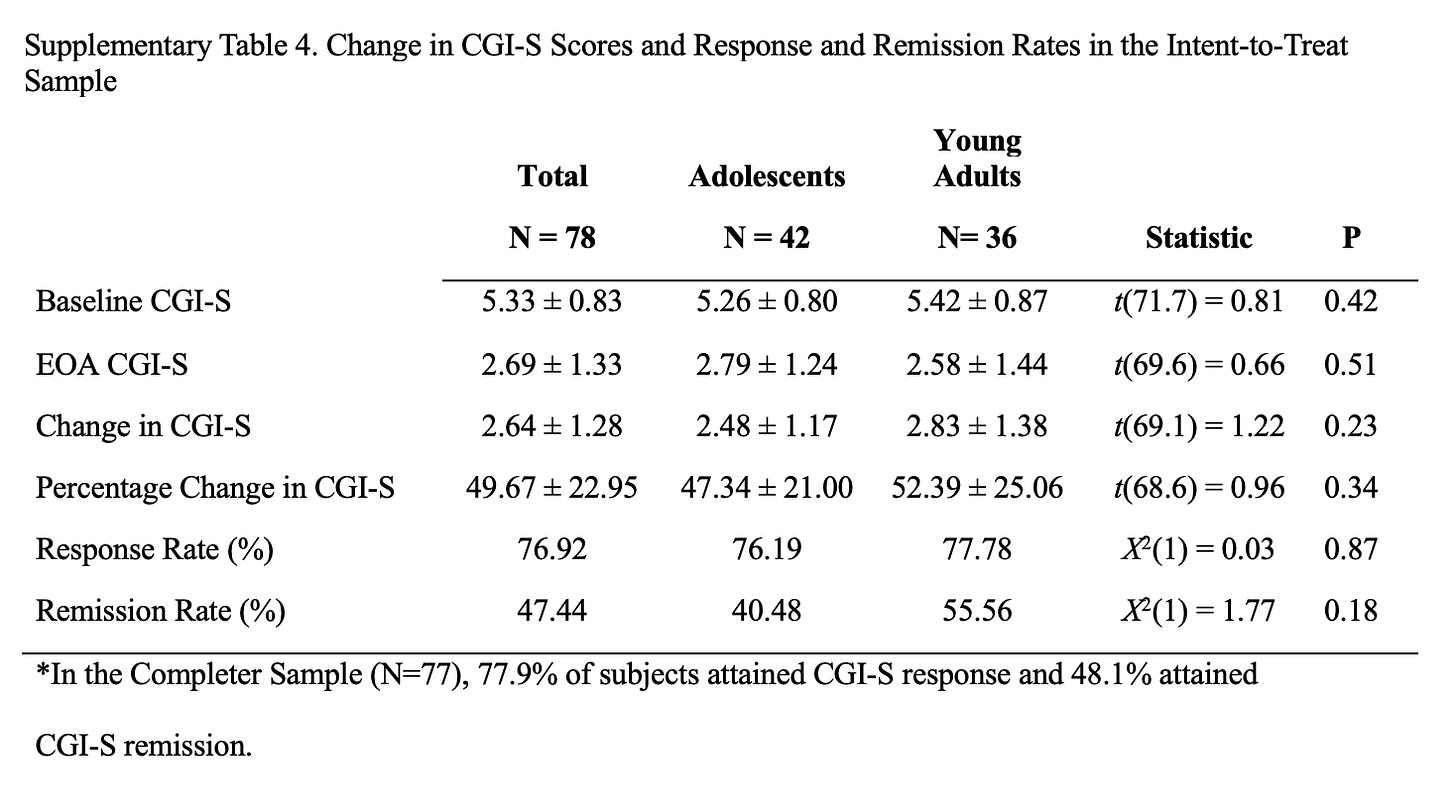
To represent these small secondary outcomes as the primary outcomes of the data sent to the FDA is incredibly deceptive. And to suggest that they were the outcomes for all 1,000 patients in the study or for “10,000 patients” who weren’t even in the study is wrong and unethical. And yet, look how TMS clinics and psychiatrists are presenting this data:
For the FDA study, the outcomes of close to 10,000 adolescents who were treated with Neurostar TMS were examined (source: Neurostar). The results showed that in those adolescents whose depression was treated with NeuroStar TMS, 78% significantly improved their depression symptoms and 48% reached complete remission. [Link, emphasis original]
Or
More than 10,000 adolescents have received over 300,000 TMS sessions worldwide, with research showing 78% experience meaningful improvement in depression symptoms and 48% reach remission.
Or:
"With an impressive 78% response rate and 48% remission rate for this population, NeuroStar is truly making a difference in the lives of adolescents across the country struggling with depression," added Fickey. [Link]
So, families are given the impression that amazing TMS outcomes have been seen in up to 10,000 “adolescents,” when it was actual 42. Just off by a factor of ~240.
4. Safety data is described, but it does not exist
In an interview in the Psychiatric Times, a psychiatrist references the FDA data to state “these are the largest study results of its kind, and they reinforce the safety and effectiveness the FDA reviewed… adverse effects were minimal.” The TMS website for WashU similarly states that the FDA data “showed the significant antidepressant benefits and safety of the treatment. Side effects were similar to those seen in adults including headaches, scalp discomfort, transient muscle twitches while under the TMS coil, fatigue, and a very small seizure risk.”
But there was zero safety data assessed in the observational study sent to the FDA. Zero. To quote the study authors themselves:
TrakStar software did not capture adverse events or side effects, and the only safety data examined here were the rate of meaningful symptomatic worsening in PHQ-9 scores.
I mean, these publications are misstating (and making up) basic factual information about this research—but reporting it with absolute confidence. It’s a bit surreal.
Where are the adults in the room to correct these inaccurate and misleading statements?
While I recognize that there’s a lot of well-intentioned excitement for TMS in adolescent depression, we have strayed so far from the evidence that I fear that families (and clinicians) are actively being misinformed.
These statements are so inaccurate that I can only come to two conclusions. Either the clinicians running these TMS clinics are so unfamiliar with the data for their treatments that they have no idea that the presented outcomes are wrong. Or, they are aware of the data and choose to present deceptive outcomes in order to increase patient volume in the clinic. The reality is that there are enormous commercial incentives to overstate the evidence for TMS in adolescents. These appear to be driving the hype around this treatment, rather than good science.
What saddens me is the silence from academics about the inaccurate and misleading public information. Aside from a single commentary, I have not come across pushback from academics against the widespread hype, errors, and deceptions. It should not be hard for our field to acknowledge that we have little high-quality data on TMS for adolescent depression. There are signals that it could be quite effective, but this is low quality data, which is what the FDA accepts for this type of device clearance. Given the urgency of adolescent depression, we really need to conduct high-quality RCTs to ensure that it is effective, especially after a large, high-quality RCT did not show that it was better than sham.
Instead, this quote continues to capture the tenor of almost all coverage:
This study marks a pivotal moment in the treatment of depression and anxiety for adolescents and young adults. It is not just about the data—it is about opening new doors for those often underserved by traditional treatments. With such a large, real-world sample, I hope both patients and providers can see the true value of TMS—particularly how NeuroStar is transforming our approach to mental health in this age group. This research represents a beacon of progress, and I believe NeuroStar will change the landscape of adolescent mental health treatment for years to come.
If this is the narrative we are going to give to poor-quality research, why bother conducting high-quality research? Why bother trying to find treatments that definitively are better than a sham treatment? It feels like a critical moment for psychiatrists to speak up about high quality versus low quality research and to push back against unjustifiable hype. And I fear that we’re failing.
Correction: an earlier version stated that the secondary CGI-S outcomes were not discussed in the article text. This was wrong. They were discussed, with the table put in the supplement. I also removed one quote about 78% of patients showing a clinically meaningful reduction in depression symptoms. Confusingly, the FDA document has outcomes that round to 78% for Individual Success Criteria, which is not clearly defined (likely a 6 point PHQ-9 score change threshold) which is at a different rate in the published paper, so this quote was ambiguous.
The biggest issues with that study? Well, only 59 adolescents were actually under 17 years-old, including 17 total 15-year olds. The vast majority of patients were 19 years or older—not true “adolescents.” Then there was the profound risk of selection bias (e.g. by excluding patients who didn’t complete PHQ-9s or who received non-standard TMS parameters) and the lack of generalizability of the sample. And finally, the dataset had almost no information about these patients—no true safety data, no information on treatment history, and no standard demographic data. You don’t have to be a genius or a cynic to recognize that this is really weak evidence.
I highlight, again this single commentary.
Throughout this piece, I’ll use “TMS for adolescent depression” or “TMS in adolescents” as shorthand for the specific FDA clearance of the NeuroStar device “as an adjunct for the treatment of Major Depressive Disorder (MDD) in adolescent patients (age 15-21).”
My charitable guess is that the authors are referencing NeuroStar used for treating 10,000 “adolescents” (whatever age range that refers to)—but this is completely different than the data submitted to the FDA and only highlights the concerns I had with that sample being unrepresentative.
Apparently 17 15-year-olds is enough data for FDA clearance…
As in my previous piece, we should be very cautious about even these response and remission rates.
And who completed at least 20 TMS sessions. These patients, coincidentally, also had significantly lower baseline PHQ-9 scores. The Clinical Global Impressions-Severity score is a summary impression from a rater about how severe the illness is on a 1-7 point scale. Literally: 1=normal, not at all ill; 2=borderline mentally ill; 3=mildly ill; 4=moderately ill; 5=markedly ill; 6=severely ill; 7=among the most extremely ill patients. That’s it!


The FDA considers 21 and under pediatric. I'll take grim satisfaction about this admittedly weak paper because I didn't co author it.